
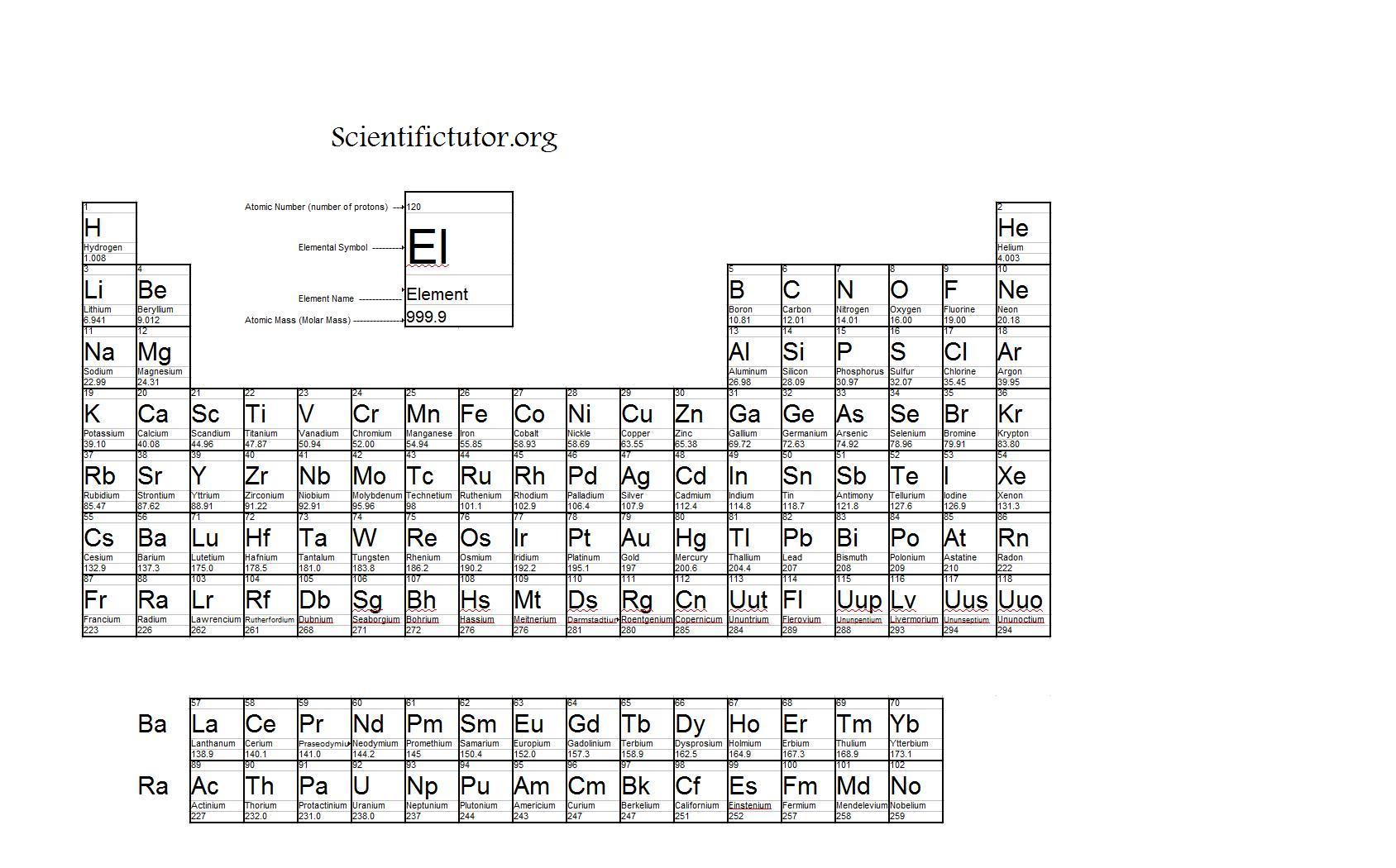
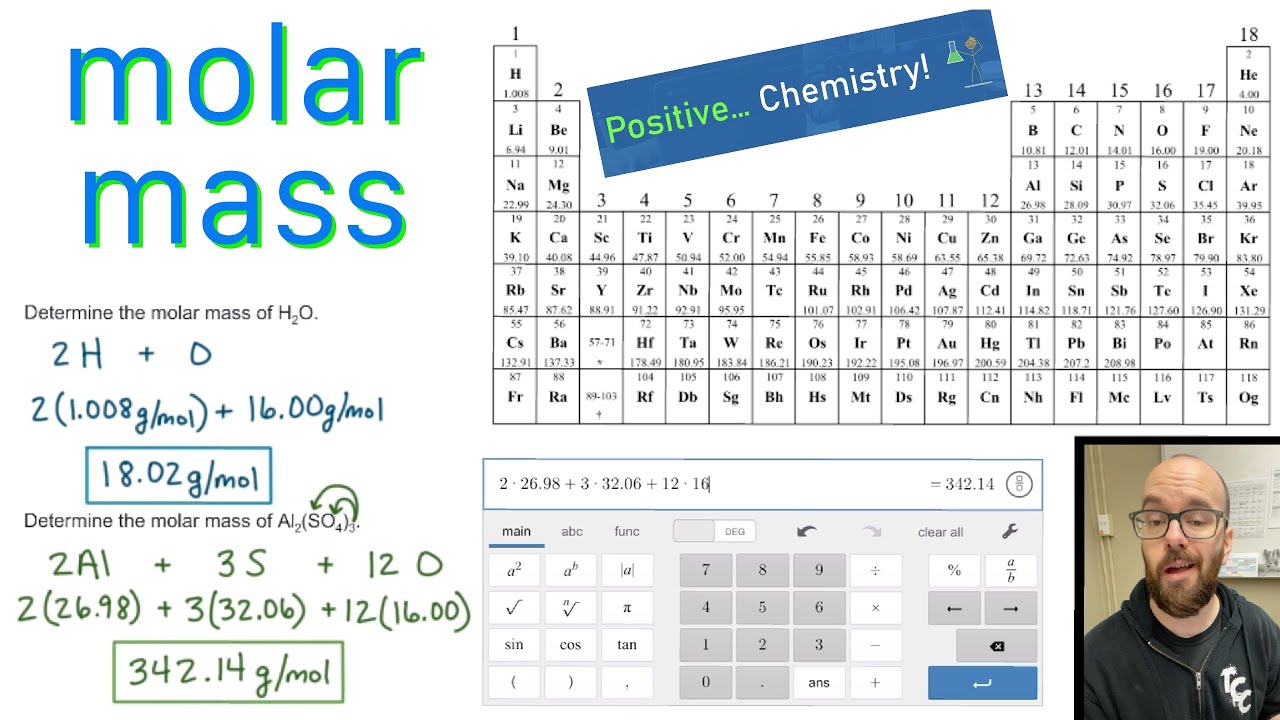
Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). The original periodic table of the elements published by Dimitri Mendeleev in 1869 arranged the elements that were known at the time in order of increasing atomic weight, since this was prior to. However when talking about a mole of an ionic compound we will still use the term molar mass. One molecule of water (H 2 O) would weigh 18.02 amu (2×1.00797 amu for H + 15.9994 amu for O), and a mole of water molecules would weigh 18.02 grams. This is because there are no individual molecules in ionic compounds. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. American Chemical Society 57-71 Lithium 6.94 Beryllium 9.012 Sodium 22.99 Magnesium 24. Periodic table-basic Created Date: 11:50:10 AM. Parenthesis indicate mass of most stable nuclide 1 2. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. Atomic masses obtained from the National Institute of Standards and Technology atomic masses with more than 5 decimal places have been rounded. The concept of molar mass can also be applied to compounds. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. concentration, fraction, ratio (molar, mass and. Rows are arranged so that elements with similar properties fall into the same vertical columns ('groups'). For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. The periodic table is a chart that organizes the elements by increasing atomic number and their chemical and physical properties (see article History of the Periodic table of elements). Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. We have defined a mole based on the isotopic atomic mass of carbon-12. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. And don’t forget to put the unit g/mol to your final calculated molar mass.) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons.This table can be obtained in PDF format here. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. First solve the brackets, then multiplications and at last do the final addition. This periodic table contains the atomic masses of the elements as accepted by the IUPAC (09-2013). The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. Always follow the calculation order to avoid any mistakes in calculation. Prepare a concept map and use the proper conversion factor.Therefore, the molar mass of Na2CO3 is 106 g/mol. If molecular formula calculator add up the total value, which is 12 + 46 + 48 106. But all these units (i.e g/mol, grams/mole and g/mole) are the same. Solution: Since sodium carbonate contains one carbon atom, two sodium atoms, and three oxygen atoms, the molecular weight is. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of KBr. Hence the Molar mass of KBr is 119.002 g/mol. So, Molar mass of KBr = Molar mass of 1 Potassium (K) atom + Molar mass of 1 Bromine (Br) atom. You can see that in KBr, there is 1 Potassium atom and 1 Bromine atom. Thus, the molar mass of bilirubin can be expressed as 584. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Now, to calculate the molar mass of KBr, you just have to add the molar mass of all the individual atoms that are present in KBr. One mole of Al atoms has a mass in grams that is numerically equivalent to the atomic mass of aluminum.
